Mucormycosis
Mucormycosis is a rare infection caused by the fungi of the order Mucorales. Normally, the human immune system is more than capable of fending molds entering our body. However, in immunocompromised patients, mucormycosis is extremely dangerous and often ends with high mortality rates. Some countries have seen a recent rise of these mycoses, most frequently in neutropenic patients with hematological diseases, ending with high mortality rates.
Concerned About Mold-Related Infections? Get a Free Inspection!
Mucormycosis is a serious mold-related infection, especially dangerous for immunocompromised individuals. Don’t take any chances with your health. Contact Mold Busters for a free virtual mold inspection. Our certified experts will provide a comprehensive assessment and actionable plan to ensure your home is safe. Act now to safeguard your health!
If you are in Montreal and suspect you might be at risk, consider our Montreal mold inspection services.
Mucormycosis causal agents

The order Mucorales holds 261 species, comprising 55 genera. Rhizopus spp., Lichtheimia spp., and Mucor spp. account for two-thirds of medically important Mucorales genera. From these, around 38 Mucorales species are connected with human infections. The most frequent mucormycosis agents are Lichtheimia corymbifera, L. ornate, L. ramose, Mucor ardhlaengiktus, M. circinelloides, M. griseocyanus, M. irregularis, M. janssenii, M. lusitanicus, Rhizopus arrhizus, R. microsporus. The most commonly isolated species worldwide is Rhizopus arrhizus (formerly R. oryzae) (1–4).

Did you know?
Only 6% of tested outdoor air samples didn’t have any presence of mold?! Find out more exciting mold stats and facts inside our mold statistics page.
Diagnosis of Mucormycosis
The most certain way to diagnose mucormycosis infection is to visualize the characteristic hyphae in tissue, sputum, exudates or scrapings, sampling, and cultivating the fungus in the laboratory. Sporangia are seldomly found, and the diagnosis is usually based on visual detection of hyphae. Mucorales fungi can be seen in infected tissues as erratically shaped, 6–25 mm in diameter (0.2–1.0 inches), non-septate, right-angle branching hyphae (Fig. 1). Non-septate hyphae with characteristic branching are the most prominent traits which prevent the misidentification of infectious Mucorales fungi with other frequent mycosis-causing species such as Aspergillus spp. or Candida spp. Mucorales species are vasculotropic fungi (attacking blood vessels), and therefore hyphal infiltrations can often be found near blood vessels of the primary infected organs.
For residents of Ottawa, our Ottawa mold inspection services can help ensure a comprehensive assessment of potential mold issues.
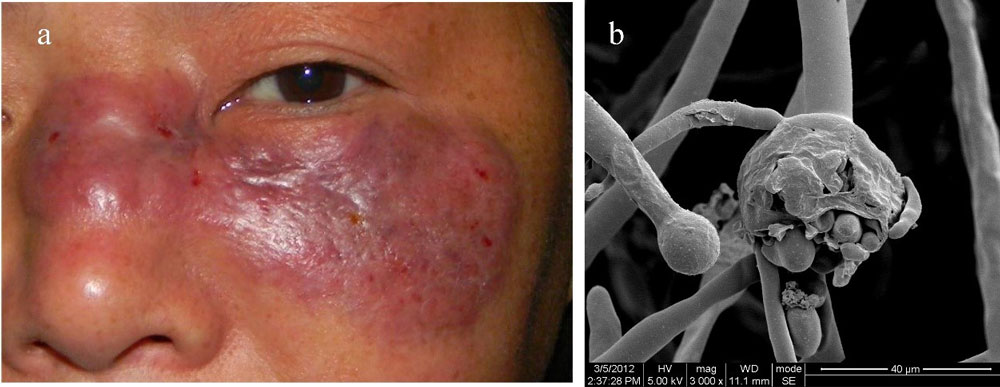
As mucormycosis is a rare disease, premortal diagnosis can be difficult. For instance, in a review study on 116 leukemia cases with pulmonary filamentous fungal infections, the correct diagnosis was made before death in only 10% percent of the patients. Similarly, in a review study of 37 hematological cancer patients with histologically documented mucormycosis, the right diagnosis was made only 35% of patients before the fatal outcomes (4–6).
Fortunately, specialized molecular tools had been developed, significantly improving the correct and timely diagnosis of this disease. The Internal transcribed spacer (ITS) region is the most widely sequenced DNA region for fungi in general. It is highly conservative within different species and can serve as a sort of a genetic barcode. It is reliable, fast, and often recommended as a first-line method for Mucorales identification (2,7).
The infection
The initial step of infection usually starts with the inhalation of fungal spores (Fig. 2). Mucormycosis can also start as wound infections and are even sometimes associated with intravenous drug abuse. Inhaled fungal spores may get deposited in the upper respiratory tract or pass through to the lung’s alveoli. Healthy organisms can fight off these intruders, as alveolar macrophages can prevent spores from germinating. However, in the immunocompromised organism, spore germination occurs, and hyphae start to grow. The formation of the mycelia (hyphal aggregate) marks the beginning of invasive mycosis. In cases of seriously reduced immunocompetence, the mycelium can invade deeper tissue layers.
This is why regular Air quality testing in your Ottawa home can be a crucial step in preventing such infections.
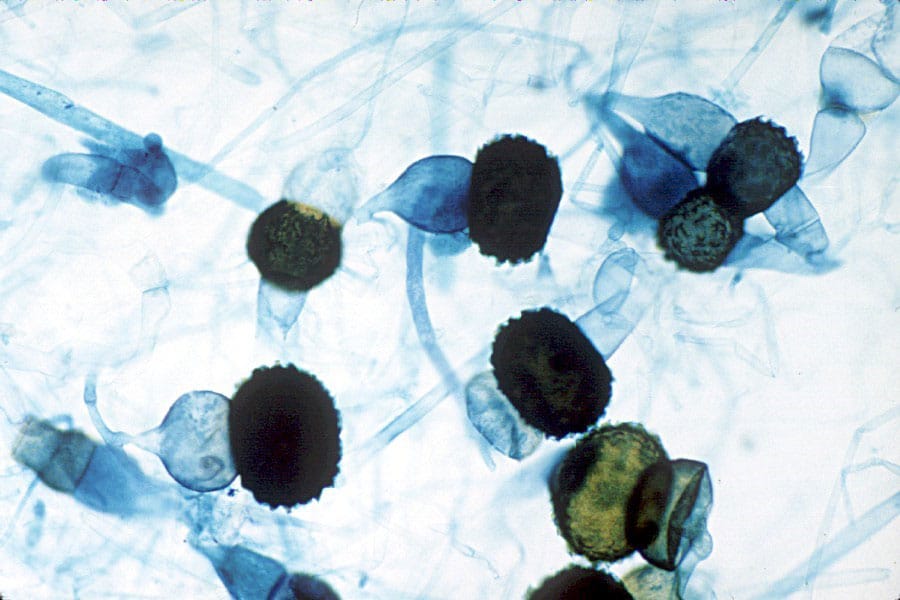
In patients suffering from pulmonary mucormycosis, symptoms often mimic clinical manifestations described for aspergillosis. The main symptoms may include cough, fever, thoracic pain, difficulty breathing, and coughing up blood. Unfortunately, infections at the initial www may often be asymptomatic (4).
Because Mucorales have a high tendency to invade blood vessels, this can often lead to the insufficient blood supply and thrombosis in the involved organ. The infections might start to spread through the bloodstream, and septic thromboembolism may appear in other organs. Because of the lacking of circulating neutrophils in the bloodstream, this often ends fatally (2,5,8).
Mucormycosis treatment
The treatment course usually stops the fungal reproduction and spreads with antifungal meds (Amphotericin B), unfortunately often with some side effects. Furthermore, Amphotericin B (either conventional or liposomal) is one of the few medications applied successfully in mucormycosis. However, if the fungus has already spread, an aggressive amputation of the infected body region must be done to stop its progress. Unfortunately, this sometimes means the removal of certain skin and bone segments, and sometimes even worse (1–3,9).
If you suspect your home may be contaminated, consider using our mold removal services.
Predisposing factors
The most important conditions that predispose to mucormycosis include (2):
- Diabetes (both with and without ketoacidosis),
- Hematological and other malignancies,
- Organ transplantation,
- Neutropenia (lower-than-normal levels of neutrophil white blood cells),
- Corticosteroid therapy (suppresses the immune system),
- Iron overload (beneficial for fungal development),
- Illicit intravenous drug use (potential direct inoculation of spores into the bloodstream),
- Neonatal prematurity,
- Malnourishment.
Regular mold testing in your Montreal home can help identify risk factors early.
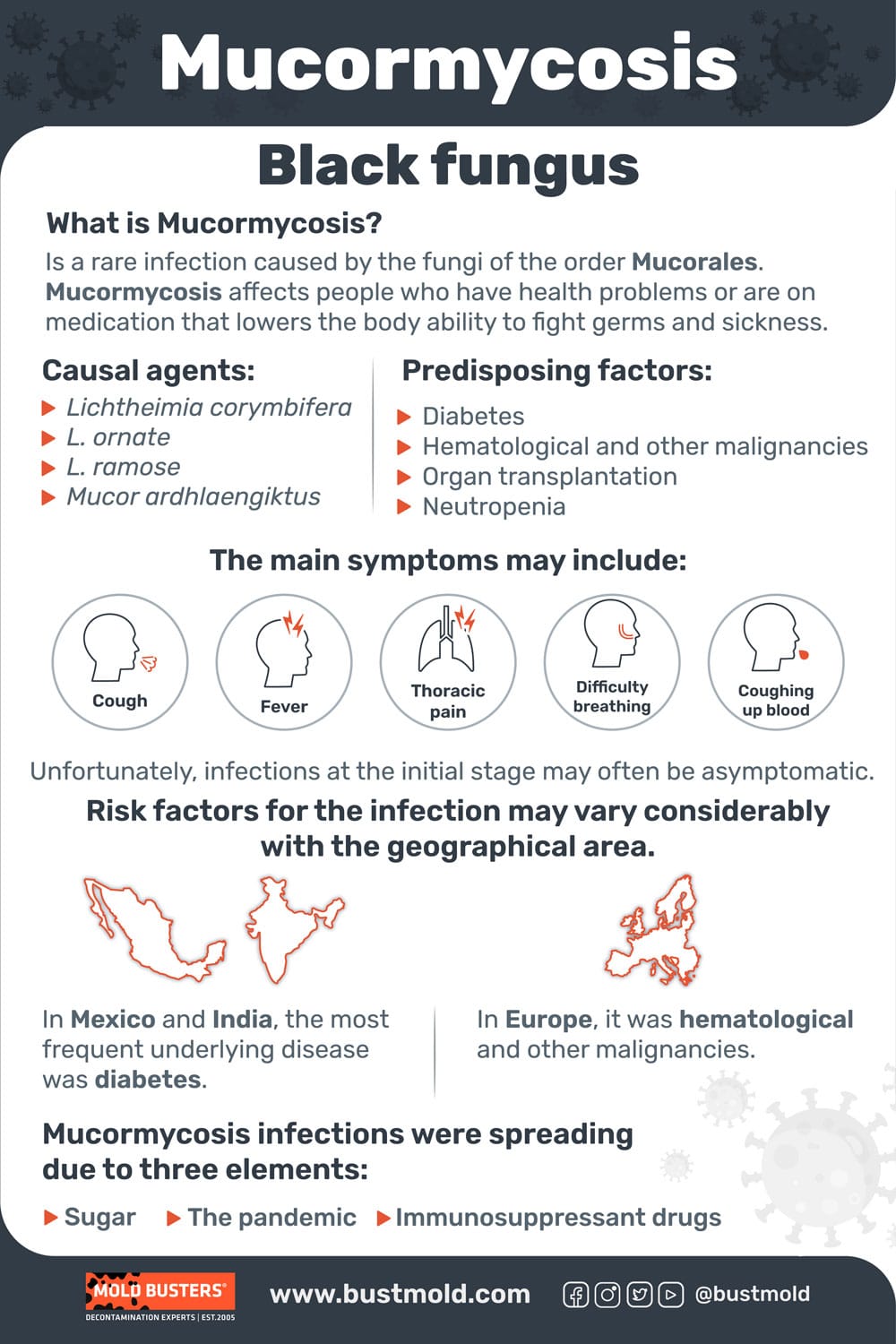
Some studies suggest that the underlying disease can be connected to the site of infection. For example, pulmonary mucormycosis can be associated with hematological malignancies and neutropenia, diabetes with sinusitis, and rhinocerebral infections, while trauma frequently leads to dermal mucormycosis. Risk factors for the infection may vary considerably with the geographical area. In Mexico and India, the most frequent underlying disease was diabetes, while in Europe, it was hematological and other malignancies (9–11).
Sometimes, several factors can come together can greatly exacerbate Mucorales infections, as it happened in 2020 in India. During the COVID-19 epidemics, a peculiar rise in several serious types of infections was noticed in already clogged hospitals of India. Swellings and headaches quickly turned to black sores spreading on throats, cheeks, eyes, and sometimes the brain. It turned out that the causal agents were of the Mucorales fungi, and the COVID-19 situation worsened the infections. According to a review study, mucormycosis infections were spreading due to three elements: sugar, immunosuppressant drugs, and the pandemic. Or, as the researchers say with their own words: “An unholy trinity of diabetes, rampant use of corticosteroid in a background of COVID-19 appears to increase mucormycosis” (9).
References:
- Jeong, W., Keighley, C., Wolfe, R., Lee, W. L., Slavin, M. A., Kong, D. C. M., & Chen, S. A. (2019). The epidemiology and clinical manifestations of mucormycosis: a systematic review and meta-analysis of case reports. Clinical Microbiology and Infection, 25(1), 26-34.
- Skiada, A., Pavleas, I., & Drogari-Apiranthitou, M. (2020). Epidemiology and diagnosis of mucormycosis: an update. Journal of Fungi, 6(4), 265.
- Walther, G., Wagner, L., & Kurzai, O. (2019). Updates on the taxonomy of Mucorales with an emphasis on clinically important taxa. Journal of Fungi, 5(4), 106.
- Eucker, J., Sezer, O., Graf, B., & Possinger, K. (2001). Mucormycoses. Mycoses, 44(7‐8), 253-260.
- Pagano, L., Ricci, P., Tonso, A., Nosari, A., Cudillo, L., Montillo, M., … & GIMEMA Infection Program. (1997). Mucormycosis in patients with haematological malignancies: a retrospective clinical study of 37 cases. British journal of haematology, 99(2), 331-336.
- Pagano, L., Ricci, P., Nosari, A., Tonso, A., Buelli, M., Montillo, M., … & Gimema Infection Program (Gruppo Italiano Malattie Ematologiche dell’Adulto). (1995). Fatal haemoptysis in pulmonary filamentous mycosis: an underevaluated cause of death in patients with acute leukaemia in haematological complete remission. A retrospective study and review of the literature. British journal of haematology, 89(3), 500-505.
- Cornely, O. A., Alastruey-Izquierdo, A., Arenz, D., Chen, S. C., Dannaoui, E., Hochhegger, B., … & Mucormycosis, E. C. M. M. (2019). Global guideline for the diagnosis and management of mucormycosis: an initiative of the European Confederation of Medical Mycology in cooperation with the Mycoses Study Group Education and Research Consortium. The Lancet infectious diseases, 19(12), e405-e421.
- Hopkins, R. J., Rothman, M., Fiore, A., & Goldblum, S. E. (1994). Cerebral mucormycosis associated with intravenous drug use: three case reports and review. Clinical infectious diseases, 19(6), 1133-1137.
- Singh, A. K., Singh, R., Joshi, S. R., & Misra, A. (2021). Mucormycosis in COVID-19: a systematic review of cases reported worldwide and in India. Diabetes & Metabolic Syndrome: Clinical Research & Reviews.
- Corzo-León, D. E., Chora-Hernández, L. D., Rodríguez-Zulueta, A. P., & Walsh, T. J. (2018). Diabetes mellitus as the major risk factor for mucormycosis in Mexico: Epidemiology, diagnosis, and outcomes of reported cases. Medical mycology, 56(1), 29-43.
- Skiada, A., Pagano, L. I. V. I. O., Groll, A., Zimmerli, S., Dupont, B., Lagrou, K., … & Petrikkos, G. (2011). Zygomycosis in Europe: analysis of 230 cases accrued by the registry of the European Confederation of Medical Mycology (ECMM) Working Group on Zygomycosis between 2005 and 2007. Clinical Microbiology and Infection, 17(12), 1859-1867.
- Yuping, R., Kaiwen, Z., Wenying, H., Jinghong, H., Xiaowei, F., Shuang, C., … & Chaoliang, Z. (2016). Observation of Fungi, Bacteria, and Parasites in Clinical Skin Samples Using Scanning Electron Microscopy. Modern Electron Microscopy in Physical and Life Sciences, 225.

Get Special Gift: Industry-Standard Mold Removal Guidelines
Download the industry-standard guidelines that Mold Busters use in their own mold removal services, including news, tips and special offers:
"*" indicates required fields
Published: October 5, 2021 Updated: July 1, 2024

Written by:
Dusan Sadikovic
Mycologist - MSc, PhD
Mold Busters
Fact checked by:
Michael Golubev
CEO
Mold Busters
