Bainier (syn. Microascus brevicaulis S.P. Abbott)
What is Scopulariopsis brevicaulis?
Scopulariopsis brevicaulis is type species of the saprophytic genus Scopulariopsis, which belongs to the family Microascaceae. It was firstly described in 1882 as Penicillium brevicaule by Saccardo and renamed Scopulariopsis brevicaulis by Bainier in 1907 [1]. It is a widespread fungus, mostly known as soil saprophytes, but it can act as an opportunistic pathogen on animals and humans [2,3]. It has proteolytic and cellulolytic abilities [4], and isolate obtained from marine sponge produces secondary metabolites scopularides A, B, and cyclodepsipeptides [5]. It has not been reported to produce mycotoxins [6], but growing on an arsenic medium can produce a poisonous compound [7]. Indoors, it can be found in dust and various damp parts of the household [2,3]. S. brevicaulis and is one of five Scopulariopsis species connected with onychomycosis, a fungal infection of the nails [8].

How does Scopulariopsis brevicaulis look like?
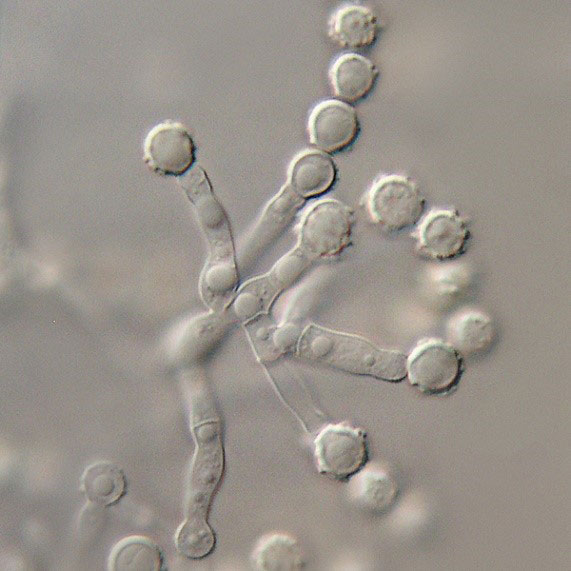
Colonies on CYA (Czapek Yeast Autolysate Agar) medium are dense and velutinous, irregularly wrinkled, and colored grey to brownish orange. It produces asexual spores – single-celled conidia borne on annelides or irregular penicillin (spore-bearing structures). Conidia are white to brown, subglobose, smooth or with ornamented texture, dimensions 6-9 x 5.5 µm [6,9] (Fig. 1). Sexual state, known as Microascus brevicaulis, was firstly discovered in 1998 by Abbott [4]. It forms black perithecia with small reniform ascospores, 5-6 µm large and 3.5-4.5 µm wide [4].
Where can Scopulariopsis brevicaulis be found?
Scopulariopsis brevicaulis can be found worldwide in soil, decaying wood, various other plant and animal products, and moist indoor environments [2,3]. It can be isolated from barley, wheat, rice grains, rice flour, soybeans, mung beans, black pepper, peanuts, groundnuts, and apples. It can also be found in nonfat dried milk, butter, cheese, salami, bacon, and luncheon meats [4,6,10]. Indoors, S.brevicaulis resides on damp walls, gypsum wallboard, and wallpaper, on wood parts, dust, as well as on fabrics and textiles [4,10].
In Ontario, Canada, S.brevicaulis was mainly isolated from mattresses and settled dust, while on walls was present in smaller amounts (3%) [10]. S. brevicaulis was the dominant species in poultry units, both indoor (40.5%) and outdoor – around poultry (62.6%) [11]. In swine, it was the second most isolated specie from indoor air and the third isolated species from swine surfaces [12]. It can act as an opportunistic pathogen on humans and animals, and its frequently isolated primarily from human skin and nails, then from animals [2,3]. Interestingly, S. brevicaulis is a parasite of ticks Dermacentor variabilis [13].
Favorable condition for Scopulariopsis brevicaulis growth
Scopulariopsis brevicaulis prefers moderate temperatures (mesophilic), with high demands for substrate water activity (aw > 0.9) [6]. S. brevicaulis grows readily in saline mediums (0.1-1 M NaCl) [13]. At 37°C (98.6°F), it can grow limitedly, while at 5°C (41°F), the growth entirely stops [6].
Is Scopulariopsis brevicaulis dangerous for humans?
S. brevicaulis is the most prevalent and significant opportunistic human pathogen from the genus Scopulariopsis[8]. It is known as a causal agent of onychomycosis and various skin problems [2]. It is found in deep cutaneous tissue of the ankle, great toe, forearm, skin, palm, fingers lip, and lymph node [8,14]. When a patient doesn’t have serious health problems, it rarely causes respiratory tract (pulmonary, bronchial) infection, sinusitis, or disseminated infections. Otherwise, S. brevicaulis can induce severe bronchial invasion. This disseminated infection affects different organs like the heart, lungs, kidneys, thorax, skin, and blood in patients with different types of leukemia [8,15]. In water-damaged homes of asthmatics’ patients in Cleveland were detected significantly higher concentrations of S. brevicaulis and Trichoderma viride than in homes without asthmatics’ [16]. Also, another dangerous characteristic of S. brevicaulis is its capability to produce toxic volatile compound trimethylarsine from the arsenic medium. This was discovered after numerous people poisoning in their homes. Wallpapers or tapestry on the walls consisted of non-volatile arsenic pigments, which fungus converted into a volatile poisonous compound with garlic odor [7].
Scopulariopsis brevicaulis causing health problems in animals
Apart from humans, S. brevicaulis are pathogenic in animals. It causes Hyperkeratosis in black calves [17], skin infections in goats and rats, and hair loss and skin lesions [18,19]. Also, it is reported tha S. brevicaulis causes rhinosinusitis in dogs [20].
Scopulariopsis brevicaulis benefits
Scientists discovered that S. brevicaulis isolated from a marine sponge produced a compound called scopularide A, significant for its specific activity against pancreatic and colon tumor cell lines [3]. Also, S. brevicaulis holds the potential to be used as a bioremediation agent. It can degrade polycyclic aromatic hydrocarbons (PAHs), the environmental pollutant that in a larger amount originates from wildfires, and combustion of other biofuels such as dung or crop residues. S. brevicaulis can degrade different forms of PAHs by 60-82% after 30 days of incubation in a liquid medium [21].
How to treat Scopulariopsis brevicaulis?
Recent studies demonstrated that S. brevicaulis is resistant to antimycotics such as amphotericin B, flucytosine, and azole compounds, so infections might not be suppressed by standard antifungal treatment [22]. Itraconazole and terbinafine were effective just in some cases regarding the S. brevicaulis caused toe onychomycosis, while griseofulvin did not show promising efficacy. Ketoconazole was partially effective but unsafe to use since it manifested some adverse effects [23]. Studies recently showed that a triple combination of caspofungin, posaconazole, and terbinafine could be promising for treating deep tissue infections caused by Scopulariopsis species [24] and treatment with 40% urea and bifonazole [25]. The use of essential oils could be an alternative solution to S. brevicaulis nail infections. In vitro studies have shown that essential oils such as cinnamon bark, geranium, clove, manuka, and thyme can inhibit the growth of S. brevicaulis and some other keratinous fungal species [26]. Also, compounds extracted from different herbs showed their antifungal potential against S. brevicaulis. Some of these compounds are thymoquinone, thymol, and thymohydroquinone from black cumin (Nigella sativa) [27], and ethyl cinnamate from the clove basil plant (Ocimum gratissimum) [28].

Did you know?
Bathrooms in Canada are the most affected by the Basidiospores mold group?! Find out more exciting mold stats and facts inside our mold statistics page.
References
- Sandoval-Denis M, Gené J, Sutton DA, Cano-Lira JF, de Hoog GS, Decock CA, Wiederhold NP, Guarro J (2016). Redefining Microascus, Scopulariopsis, and allied genera. Persoonia 36: 1–36.
- Park J, Kwon W, Hong S-B, Han K-H (2020). First Record of the Complete Mitochondrial Genome of a Saprotrophic and Opportunistic Human Pathogenic Fungus, Scopulariopsis brevicaulis. Mycobiology, 48 (6): 528-531.
- Lukassen MB, Saei W, Sondergaard TE, Tamminen A, Kumar A, Kempken F, Wiebe MG, Sørensen JL (2015). Identification of the Scopularide Biosynthetic Gene Cluster in Scopulariopsis brevicaulis. Marine drugs, 13: 4331-4343.
- Abbott SP, Sigler L, Currah RS (1998). Microascus brevicaulis sp. nov., the Teleomorph of Scopulariopsis brevicaulis. Supports Placement of Scopulariopsis with the Microascaceae. Mycologia, 90 (2): 297-302.
- Yu Z, Lang G, Kajahn I, Schmaljohann R, Imhoff JF (2008). Scopularides A and B, Cyclodepsipeptides from a Marine Sponge-Derived Fungus, Scopulariopsis brevicaulis. Journal of Natural Products, 71 (6): 1052–1054.
- Pitt JI, Hocking AD (2009). Fungi and food spoilage. New York, NY: Springer, pp 72,73.
- Cox DP (1975). Microbiological Methylation of Arsenic. In Arsenical Pesticides (Woolson ed.). American Chemical Society, pp. 81-96.
- Iwen PC, Schutte SD, Florescu DF, Noel-Hurst RK, Sigler L (2012). Invasive Scopulariopsis brevicaulis infection in an immunocompromised patient and review of prior cases caused by Scopulariopsis and Microascus species. Medical Mycology, 50: 561–569.
- Abbott SP, Sigler L (2001). Heterothallism in the Microascaceae Demonstrated by Three Species in the Scopulariopsis brevicaulis. Mycologia, 93 (6): 1211-1220.
- Flannigan B, Miller JD (2011). Microbial growth in indoor environments. In Microorganisms in Home and Indoor Work Environments Diversity, Health Impacts, Investigation and Control (Flannigan et al. eds.). Taylor & Francis Group, pp.75.
- Viegas C, Viegas S, Monteiro A, Carolino E, Sabino R, Veríssimo C (2012). Comparison of indoor and outdoor fungi and particles in poultry units. In Environmental impact (Brebbia and Chon eds). Wessex: WIT Press, pp. 589-96.
- Viegas C, Carolino E, , Sabino R, Viegas S, Veríssimo C (2013). Fungal Contamination in Swine: A Potential Occupational Health Threat. Journal of Toxicology and Environmental Health, Part A, 76:272–280.
- Yoder JA, Benoit JB, Zettler LW (2009). Moisture requirements of a soil imperfect fungus, Scopulariopsis brevicaulis Bainier, in relation to its tick host. International Journal of Acarology 29 (3): 271-277.
- Petanović M, Tomić Paradžik M, Krištof Ž, Cvitković A, Topolovac Z (2010). Scopulariopsis brevicaulis as the Cause of Dermatomycosis. Acta Dermatovenerologica Croatica,18(1):8-13.
- Yang Q, Wei J, Chen Z (2012). Fatal bronchial invasion of Scopulariopsis brevicaulis in an acute monocytic leukemia patient. Diagnostic Microbiology and Infectious Disease, 73(4):369-71.
- Vesper SJ, McKinstry C, Yang C, Haugland RA, Kercsmar CM, Yike I, Schluchter MD, Kirchner HL, Sobolewski J, Allan TM, Dearborn DG (2006). Specific Molds Associated With Asthma in Water-Damaged Homes. Journal of Occupational and Environmental Medicine, 48(8):852-8.
- Ogawa S, Shibaharay T, Sanoz A, Kadotax K, Kuboy M (2008). Generalized Hyperkeratosis Caused by Scopulariopsis brevicaulis in a Japanese Black Calf. Journal of Comparative Pathology, 138(2-3):145-50.
- Ozturk D, Adanir R, Turutoglu H (2009). Superficial skin infection with Scopulariopsis brevicaulis in two goats. A case report. Bulletin of the Veterinary Institute in Puławy, 53 (3): 361-363.
- Yapıcıer OS, Kaya M, Erol Z, Öztürk D (2020).Isolation of Scopulariopsis brevicaulis from Wistar Rats. Etlik Vet Mikrobiyol Derg, 31(2): 196-200.
- Sri-Jayantha L, Matthews KG, Scharf V (2019). Scopulariopsis brevicaulis Rhinosinal Infection in a Dog. Journal of the American Animal Hospital Association 55(1): e551-02.
- Mao J, Guan W (2016). Fungal degradation of polycyclic aromatic hydrocarbons (PAHs) by Scopulariopsis brevicaulis and its application in bioremediation of PAH-contaminated soil. Acta Agriculturae Scandinavica, Section B — Soil & Plant Science, 66 (5):1-7.
- Skora M, Macura AB, Bulanda M (2014). In vitro antifungal susceptibility of Scopulariopsis brevicaulis isolates. Medical Mycology, 52: 723–727.
- Gupta AK, Gregurek-Novak T (2001). Efficacy of Itraconazole, Terbinafine, Fluconazole, Griseofulvin and Ketoconazole in the Treatment of Scopulariopsis brevicaulis Causing Onychomycosis of the Toes. Dermatology, 202(3):235-8.
- Yao L, Wan Z, Li R, Yu J (2015). In Vitro Triple Combination of Antifungal Drugs against Clinical Scopulariopsis and Microascus Species. Antimicrobial Agents and Chemotherapy, 59 (8): 5040-5043.
- Piraccini BM, Bruni F, Alessandrini A, Starace M (2015). Evaluation of efficacy and tolerability of four weeks bifonazole treatment after nail ablation with 40% urea in mild to moderate distal subungual onychomycosis. Dermatologia e Sifilografia, 151(1):32-36.
- Michalczyk A, Ostrowska P (2021). Essential oils and their components in combating fungal pathogens of animal and human skin. Journal of Medical Mycology 31(2):101118.
- El-Hack MEA, Alagawany M, Farag MR, Tiwari R, Karthik K, Dhama K (2016). Nutritional, Healthical and Therapeutic Efficacy of Black Cumin (Nigella sativa) in Animals, Poultry and Humans International Journal of Pharmacology, 12: 232-248.
- Dubey NK, Tiwari TN, Mandin D, Andriamboavonjy H, Chaumont J-P (2000). Antifungal properties of Ocimum gratissimum essential oil ethyl cinnamate chemotype. Fitoterapia, 71:567-569.
- Featured image photo, A Hydroxypropyl Chitosan (HPCH) Based Medical Device Prevents Fungal Infections: Evidences from an In Vitro Human Nail Model – Scientific Figure on ResearchGate.

Get Special Gift: Industry-Standard Mold Removal Guidelines
Download the industry-standard guidelines that Mold Busters use in their own mold removal services, including news, tips and special offers:

Written by:
Aleksandra Zebeljan
Mycologist
Mold Busters
Edited by:
Dusan Sadikovic
Mycologist – MSc, PhD
Mold Busters
