G.A. Huber 1933
What is Aspergillus sclerotiorum?
Aspergillus sclerotiorum is a filamentous, asexual fungus belonging to the genus Aspergillus section circumdati. It is a saprophyte often found in soil and as a stored foods contaminant. It is characteristic for the formation of colonies with sclerotia, rarely an opportunistic pathogen, and it produces low amounts of several mycotoxins. A. sclerotiorum also has the potential in biocontrol, as it is pathogenic to some termite species (1,2).

How does Aspergillus sclerotiorum look like?
A morphological characteristic of A. sclerotiorum is that it forms colonies dominated by white to cream sclerotia areas, with light cream to yellow sporulation (Fig. 1). Generally, sclerotia give the fungus the ability to safely endure harsher conditions. Furthermore, sclerotium is considered a significant requirement for sexual development in Aspergillus (section Circumdati), even though the sexual state of A. sclerotiorum has not been yet identified. However, sclerotia in aspergilli may also serve as a repository for cleistothecia (2–4).
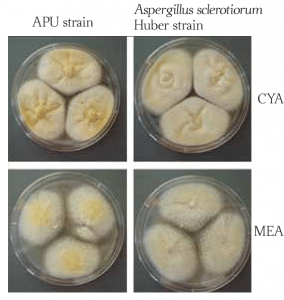
Morphologically, A. sclerotiorum colonies grown on CYA and MEA are very similar. The surface of the colonies is floccose (covered with woolly tufts), with white mycelial areas. Sporulation is cream to light yellow. CYA-grown (Czapek Yeast Autolysate Agar) colonies produce a yellow soluble pigment, and a clear exudate, while MEA-grown (Malt Extract Agar) colonies lack both pigments and exudation. The CYA colonies reverse light yellow greyish-yellow, while the MEA reverse is brown (2). Growing optimum temperature lies between 25–30 °C (77–86°F).
Conidiophores of A. sclerotiorum are biseriate structures with globose vesicles. Biseriate entails that both metulae and phialides are present and are formed simultaneously. Metulae and phialides are structures holding the conidia, and Metulae cover 75–100 % of the vesicle. Conidia are globose to subglobose, smooth-walled, 2–3.5 μm in diameter (2).
Is Aspergillus sclerotiorum toxic?
Aspergillus sclerotiorum is known to produce a mycotoxin called ochratoxin. Ochratoxins are polyketide derivatives and carcinogenic fungal secondary metabolites. Polyketides are among the most plentiful fungal secondary metabolites. They are complex molecules with continuous use in the pharmaceutical industry to manufacture antibiotics such as doxycycline and erythromycin.
Various fungal species are known to produce ochratoxins, however not all of them with consistency. A. sclerotiorum strains have not shown consistency in ochratoxin production, and when they do, the mycotoxin was produced in low amounts (2). Nevertheless, ochratoxins can contaminate stored grains, legumes, coffee, dried fruits, alcoholic beverages, and meat products. They can harm the kidneys, cause tumors, raise the risk of birth defects, and damage the immune system of rats and, thusly, likely of humans (5–7).
Some A sclerotiorum strains also produce xanthomegnin, viomellein, and vioxanthin mycotoxins, which can be harmful to animals and humans. This species can also produce weak amounts of penicillic acid, a major product of acid degradation of penicillin (2,8).
What are the health risks of Aspergillus sclerotiorum?
Aspergillus sclerotiorum as a food contaminant may be a health risk due to mycotoxin production, but it can also act as an opportunistic pathogen in some instances. Otomycosis, also known as fungal otitis externa, is the disease of the other parts of the ear canal. This disease is usually caused by A. niger, A. fumigatus, and A. flavus. However, A. sclerotiorum can cause this infection as well. This was recorded in a 46-year-old Japanese man, admitted with discomfort and discharge in his right ear. The cause turned out to be A. sclerotiorum infection of the external ear canal.
Furthermore, as it is characteristic for otomycosis, the patient has undergone mastoidectomy fifteen years before this infection. Mastoidectomy is a procedure where tissue is removed from the hollow spaces in the skull, behind the ear, within the mastoid bone. It is speculated that the excess of cerumen (earwax) had created a suitable condition for this fungus to spread. In the ear canal, A. sclerotiorum forms whitish, fluffy, cotton-like mycelium, which can be found together with keratinous debris and exudates. Otomycosis can also be stimulated by the use of broad-spectrum antibiotics and corticosteroids therapy.
A. sclerotiorum can also cause onychomycosis, typically caused by keratinous fungi such as Epidermophyton, Microsporum, and Trichophyton. Onychomycosis is an infection of nails by a fungus that feeds on the protein keratin found in human skin, nails, and hair (9–12). This species is considered as a species characteristic for homes with water damage and is thusly listed in the Group 1 of the ERMI (The Environmental Relative Moldiness Index) list of potentially hazardous fungi (13).

Did you know?
Chaetomium is the 2nd common toxic mold type found in homes we tested?! Find out more exciting mold stats and facts inside our mold statistics page.
References
- Hansen, G. M., Laird, T. S., Woertz, E., Ojala, D., Glanzer, D., Ring, K., & Richart, S. M. (2016). Aspergillus sclerotiorum fungus is lethal to both Western drywood (Incisitermes minor) and Western subterranean (Reticulitermes hesperus) termites.
- Visagie, C. M., Varga, J., Houbraken, J., Meijer, M., KocsubÚ, S., Yilmaz, N., … & Samson, R. A. (2014). Ochratoxin production and taxonomy of the yellow aspergilli (Aspergillus section Circumdati). Studies in mycology, 78, 1-61.
- Dyer, P. S., & O’Gorman, C. M. (2012). Sexual development and cryptic sexuality in fungi: insights from Aspergillus species. FEMS microbiology reviews, 36(1), 165-192.
- Frisvad, J. C., Petersen, L. M., Lyhne, E. K., & Larsen, T. O. (2014). Formation of sclerotia and production of indoloterpenes by Aspergillus niger and other species in section Nigri. PLoS One, 9(4), e94857.
- Abdel-Azeem AM, Abdel-Azeem MA, Abdul-Hadi SY, Darwish AG. Aspergillus: Biodiversity, Ecological Significances, and Industrial Applications. 2019;121–79.
- Tsang, C. C., Tang, J. Y., Lau, S. K., & Woo, P. C. (2018). Taxonomy and evolution of Aspergillus, Penicillium and Talaromyces in the omics era–Past, present and future. Computational and Structural Biotechnology Journal, 16, 197-210.
- Bayman, P., Baker, J. L., Doster, M. A., Michailides, T. J., & Mahoney, N. E. (2002). Ochratoxin production by the Aspergillus ochraceus group and Aspergillus alliaceus. Applied and environmental microbiology, 68(5), 2326-2329.
- Frisvad, J. C., Frank, J. M., Houbraken, J. A. M. P., Kuijpers, A. F., & Samson, R. A. (2004). New ochratoxin A producing species of Aspergillus section Circumdati. Studies in mycology, 50(1), 23-43.
- Miertusova, S., & Simaljakova, M. (2003). Yeasts and fungi isolated at the mycology laboratory of the First Dermatovenerology Clinic of the Medical Faculty Hospital of Comenius University in Bratislava 1995-2000. Epidemiologie, mikrobiologie, imunologie: casopis Spolecnosti pro epidemiologii a mikrobiologii Ceske lekarske spolecnosti JE Purkyne, 52(2), 76-80.
- Anwar, K., & Gohar, M. S. (2014). Otomycosis; clinical features, predisposing factors and treatment implications. Pakistan journal of medical sciences, 30(3), 564.
- Harima, N., Inoue, T., Kubota, T., Okada, O., Ansai, S. I., Manabe, M., … & Kasai, T. (2004). A case of otomycosis caused by Aspergillus sclerotiorum. The Journal of dermatology, 31(11), 949-950.
- Singh, S. M., & Barde, A. K. (1983). A case of onychomycosis caused by Aspergillus sclerotiorum. Indian journal of dermatology, venereology and leprology, 49(1), 22-25.
- Vesper, S., McKinstry, C., Haugland, R., Wymer, L., Bradham, K., Ashley, P., … & Friedman, W. (2007). Development of an environmental relative moldiness index for US homes. Journal of Occupational and Environmental Medicine, 49(8), 829-833.
- Featured photo, Mikael Rørdam Andersen at the group Network Engineering in Eukaryotic Cell Factories at Technical University of Denmark in Denmark., Assessed from Wikipedia.

Get Special Gift: Industry-Standard Mold Removal Guidelines
Download the industry-standard guidelines that Mold Busters use in their own mold removal services, including news, tips and special offers:

Written by:
Dusan Sadikovic
Mycologist – MSc, PhD
Mold Busters
Fact checked by:
Michael Golubev
General Manager
Mold Busters
